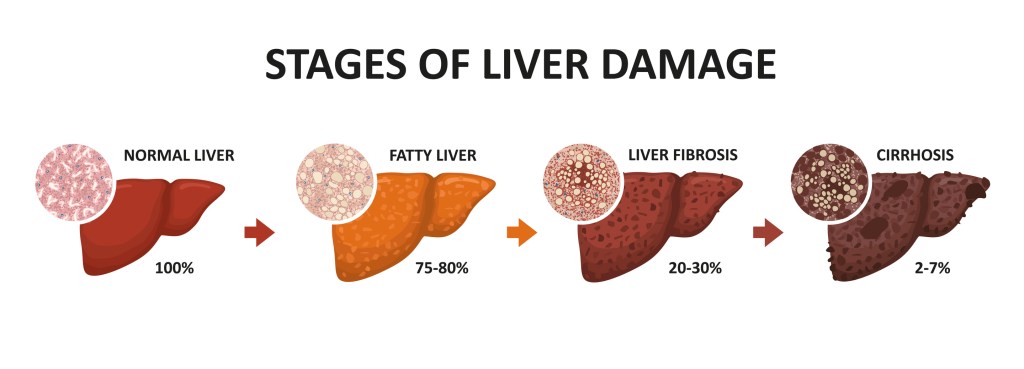
DiabetesDaily informed me of a new drug available for treatment of a liver disease that affects “up to 20% of people with diabetes. The disease is MASH: metabolic dysfunction-associated steatohepatitis, something else I’d never heard of. (Shouldn’t the acronym be MDASH?) The drug is resmetirom, sold in the U.S. as Rezdiffra. Click for the FDA announcement. MASH can lead to liver scarring (fibrosis), which then qualifies the patient for resmetirom. The DiabetesDaily article is well-written and includes alternatives to this new drug.
I’ve long been aware of NASH: non-alcoholic steatohepatitis). THIS is the liver disease target according to the FDA announcement, which states “Rezdiffra is a partial activator of a thyroid hormone receptor; activation of this receptor by Rezdiffra in the liver reduces liver fat accumulation.” Furthermore:
The most common side effects of Rezdiffra included diarrhea and nausea. Rezdiffra comes with certain warnings and precautions, such as drug-induced liver toxicity and gallbladder-related side effects.
There may be potential significant interaction of this new drug with others, particularly statin cholesterol-lowering drugs.
Color me skeptical. If only because the drug was on the “accelerated approval pathway.” But I’ll keep and open mind.
I’m not sure, but it appears that candidates for the drug will need a liver biopsy showing fibrosis (scarring).
Steve Parker, M.D.